WHA’s Hydrogen Fire Risk Management Philosophy
Highlights
- Hydrogen has seen increased use over the past decade — but also an increased risk of incidents.
- Hydrogen can be used safely when appropriate safety considerations are applied.
- WHA employs a risk management approach focused on prevention, anticipating ignition, and adopting best practices to mitigate hydrogen fire risks.
Hydrogen is a valuable resource with incredible potential. Over the past decade, industry has seen a rise in the use of hydrogen technologies. However, with increased use comes an increased risk of incidents.
Although some think of hydrogen as unsafe, it has actually been used safely across a variety of industries for generations. Some of hydrogen’s properties make it safer to handle and use than many of today’s common fuels. Of course, other properties of hydrogen require special engineering considerations to prevent an accident.
The first step to safety is understanding. Read on to discover or refresh your knowledge about the unique hazards of hydrogen, and learn more about WHA’s hydrogen fire risk analysis approach.

Our Hydrogen Safety Knowledge is Built on Experience
WHA’s hydrogen safety expertise is informed by years of failure analysis and forensic engineering. Our engineers have investigated numerous incidents involving hydrogen and other hazardous fuels. The incident pictured above, for example, occurred at a power plant in Muskingum River in 2007.
This tragic incident resulted from the release of hydrogen from a failed rupture disk on a storage vessel. A large quantity of hydrogen collected under a roof and ignited, killing one and injuring 10 others.
It’s our mission to prevent incidents like this. We have developed our suite of hydrogen consulting services and resources to help keep industry safe.

Why Hydrogen Presents a Serious Fire Hazard
So how does a hydrogen fire occur? We like to explain this by using the illustration of the “fire triangle.”
There are generally three factors essential to all fires: fuel, oxidizer, and ignition. What we call fire is actually a “combustion” reaction: the chemical process of a fuel like hydrogen reacting with an oxizider like oxygen. Under controlled circumstances, this powerful reaction can be harnessed for good. Under the wrong circumstances, a volatile hydrogen fire can be extremely destructive.! Let’s review the properties of hydrogen that make it unique and crucial to handle safely.
Safety Challenge #1: Hydrogen is Challenging to Detect and Contain
Hydrogen is a colorless, odorless, tasteless gas, and when it burns, it emits very little light and color. This makes hydrogen and hydrogen fires very difficult to detect without special equipment.
Hydrogen also holds a familiar place as the smallest element on the periodic table. In application, this means that its tiny H2 molecules are very susceptible to leaks. They can diffuse through many materials considered to be “airtight.” Hydrogen can even absorb into certain metals, causing them to become brittle and crack (known as hydrogen “embrittlement”).
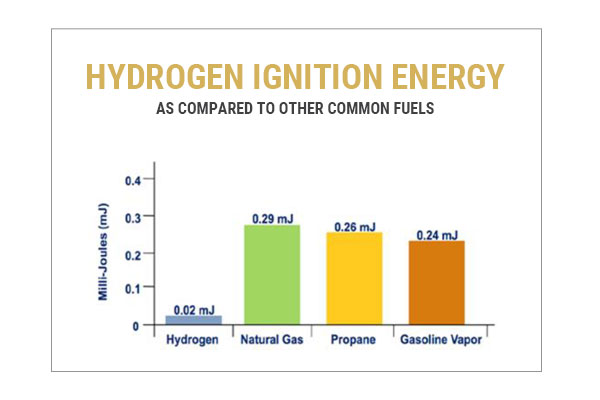
Safety Challenge #2: Hydrogen Requires a Low Ignition Energy
When hydrogen mixes with an oxidizer, it’s extremely easy to ignite. Gasoline, for instance, requires about 0.24 mJ of energy to ignite under the right conditions in air. This is less energy than generated from a small static discharge from your finger. By comparison, hydrogen only requires about 0.02mJ, 12 times less ignition energy than gasoline! You can probably imagine all the energy sources that are therefore capable of igniting hydrogen mixtures. This means that it’s a dangerous situation if hydrogen finds its way out of a contained system.
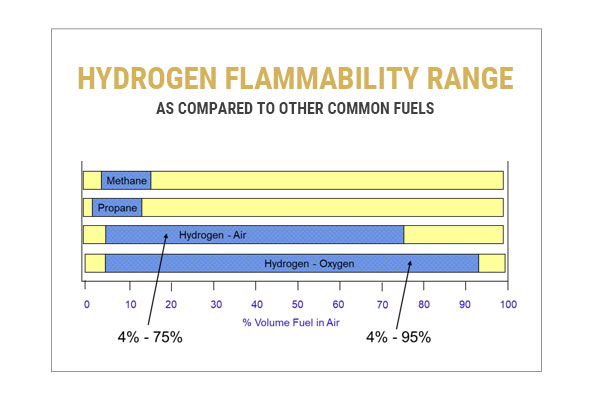
Safety Challenge #3: Hydrogen Has a Wide Flammability Range
All fuels are only flammable when mixed in an appropriate ratio with an oxidizer, known as a flammability range. Unlike common hydrocarbon fuels such as methane and propane, hydrogen has a very wide flammability range. It is vulnerable to ignition anywhere from 4-75% in air (and up to 95% in pure oxygen). This means that extra care must be taken to prevent leaks in hydrogen systems.
WHA’s Solution: Hydrogen Risk Management Approach
To reduce the risk of fire, WHA engineers implement an approach based on three simple concepts: Avoid, Anticipate, and Adopt. These three concepts address each of the three legs of the fire triangle. By implementing this strategy, we minimize the chance that all three fire factors are present at the same time and can help prevent hydrogen fires.
Concept 1: Avoid Unintentional Mixing
By preventing accidental release and internal leaks in your hydrogen system, contained hydrogen remains in a concentration range which is not flammable and poses no fire risk. Design and material selection are of utmost importance for safe hydrogen systems.
Concept 2: Anticipate Ignition Mechanisms
With a very low minimum ignition energy required, ignition can occur readily from a wide variety of sources (e.g. electrostatic discharge, metal fracture, friction, shock waves, and even light). Although care must be taken to prevent ignition, users should assume an ignition source is always present when a combustible hydrogen-oxidizer mixture has formed.
Concept 3: Adopt best practices
By implementing best practices for design, inspection, assembly, operation, maintenance, and safety systems, the risk of a hydrogen fire can be greatly reduced.

Hydrogen Safety Summarized
Yes, hydrogen can be hazardous. While it’s not toxic, it’s flammable and can catch fire easily. It’s also hard to detect because it’s colorless and odorless. This means hydrogen needs careful handling to avoid accidents. That being said, hydrogen has been used safely in many settings for decades and can be safe with proper precautions and personnel training.
Yes, hydrogen is extremely flammable. It can catch fire easily when mixed with air and exposed to an ignition source like a spark. This is because it has a wide range of concentrations where it can burn and a low ignition energy.
Hydrogen is hazardous because it’s difficult to detect and contain and can ignite easily at a wide range of concentrations.
Hydrogen is incredibly useful and has many applications in industry and beyond. It will also likely play a crucial role as an alternative fuel in the clean energy transition. With proper preventative measures, hydrogen can be used safely, and it is naturally safer to use than other fuels in some respects.
Anyone who uses or works around hydrogen gas must be provided proper safety training and take hydrogen safety seriously. Executives and managers in industry must follow international safety guidelines and undergo hydrogen risk assessment to ensure public and personnel safety.
1. WHA’s hydrogen safety training
2. WHA’s hydrogen articles and case studies
3. H2 Tools (Lessons learned, Best safety practices, Materials database, and Safety Planning)
4. ANSI/AIAA G-095A: Guide to Safety of Hydrogen and Hydrogen Systems
5. ISO/TR 15916: Basic Considerations for the Safety of Hydrogen Systems
6. CGA G-5-2017: Hydrogen

Next Steps to Prevent Hydrogen Fire: Safety Measures
WHA is dedicated to helping industry use hydrogen safely. Our specialized engineers are uniquely positioned to equip your staff with the tools and knowledge necessary to create and maintain a safe environment for everyone.
- Training: WHA provides web-based and on-site hydrogen safety training, custom-tailored for your unique application and needs.
- Hazard Analysis: WHA’s Hydrogen Combustion Risk Analysis (HCRA) approach and expertise can be applied to assess component and system level hazards in your application.
- Testing: With both a laboratory test facility and WHA’s Proving Grounds for large or high-energy tests, WHA provides custom and standard testing as well as research services.
Originally published November 18, 2019.
Share this entry
Related Articles
What New CSA Group Research Reveals about Hydrogen Leak Testing Gaps
How do we reliably detect, measure, and manage hydrogen leaks? A new CSA Group research study takes…
Case Study: Recreating High-Energy Cylinder Ruptures at Full Scale
Several years ago, personnel were unloading a high-pressure aluminum cylinder containing a reactive synthesis gas blend when…
WHA Failure Analysis Legacy: Lessons Learned in Forensic Engineering
What are some of the biggest lessons you’ve learned through forensic engineering? Brad Forsyth: Our failure analysis…
Request an expert
consultation
Contact us to request a free consultation with an experienced engineer who can help you better understand your needs and our solutions.



