Oxygen compatibility and safe-use criteria
Because materials ignite and burn more easily in oxygen-enriched atmospheres, it is important to determine how “compatible” materials are for oxygen service.
Once ignited, even small gaskets, O-rings, or other nonmetals in oxygen components can start a “kindling chain” reaction that leads to ignition of surrounding materials, including metals. For this reason, we often place a special emphasis on qualifying nonmetal materials for use with oxygen.
Oxygen component designers often look for lists of definitive “safe-use” criteria, but unfortunately the issue of oxygen compatibility is not so black and white. Safe-use criteria are not determined by any single test, but by an overall analysis of ignition probability and consequence in the application.
In recent years, industry experts have increasingly observed that common misperceptions about safe-use have put human lives and equipment at risk. Failure to understand the principles of oxygen compatibility can lead to dangerous oxygen fires.
In this article, WHA’s oxygen experts will guide us through the basic science of oxygen compatibility. We will take a look at how we qualify materials for use in oxygen, incorporating industry guides, standard tests, and fire risk analysis.
For a closer look at the topics discussed, keep an eye out for several papers to be published through ASTM international later this year (more on that below).
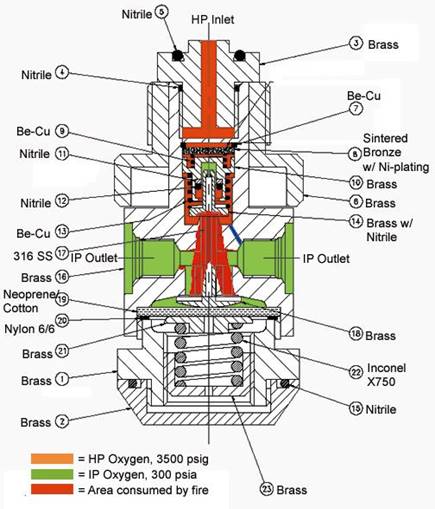
What is oxygen compatibility?
In order to characterize oxygen compatibility, we look to industry standards published through various organizations, including ASTM International, International Standards Organization (ISO), National Aeronautics and Space Administration (NASA), European Norm (EN), and British Standards Institute (BSI).
In every WHA oxygen safety training course, we first reference ASTM G126 for a broadly accepted definition of oxygen compatibility:
Oxygen Compatibility (n): “The ability of a material to coexist at an expected pressure and temperature with both oxygen and a potential source of ignition.”
In other words, a material is safe for use with oxygen only if it can successfully perform its design and function under the expected conditions (like pressure, concentration, heat, and presence of other ignition sources) with a low probability of ignition and fire.
It is important to note that because a material’s oxygen compatibility depends heavily on the severity of each side of the fire triangle: oxidizer (oxygen use conditions), fuel (size/geometry of the material), and ignition source (energy associated with the application), oxygen compatibility for a material is specific to an application and can change if one or more of these conditions change. Thus, the term “safe-use” for materials in oxygen is both relative and application-specific.

How do we characterize oxygen compatibility?
Several respected laboratories around the world, including WHA International, can perform standardized tests provided by international organizations to help characterize oxygen compatibility. Methodical collection, comparison, and analysis of test data reveals important information about how materials should — or should not — be used with oxygen.
These tests fall under one of three primary types:
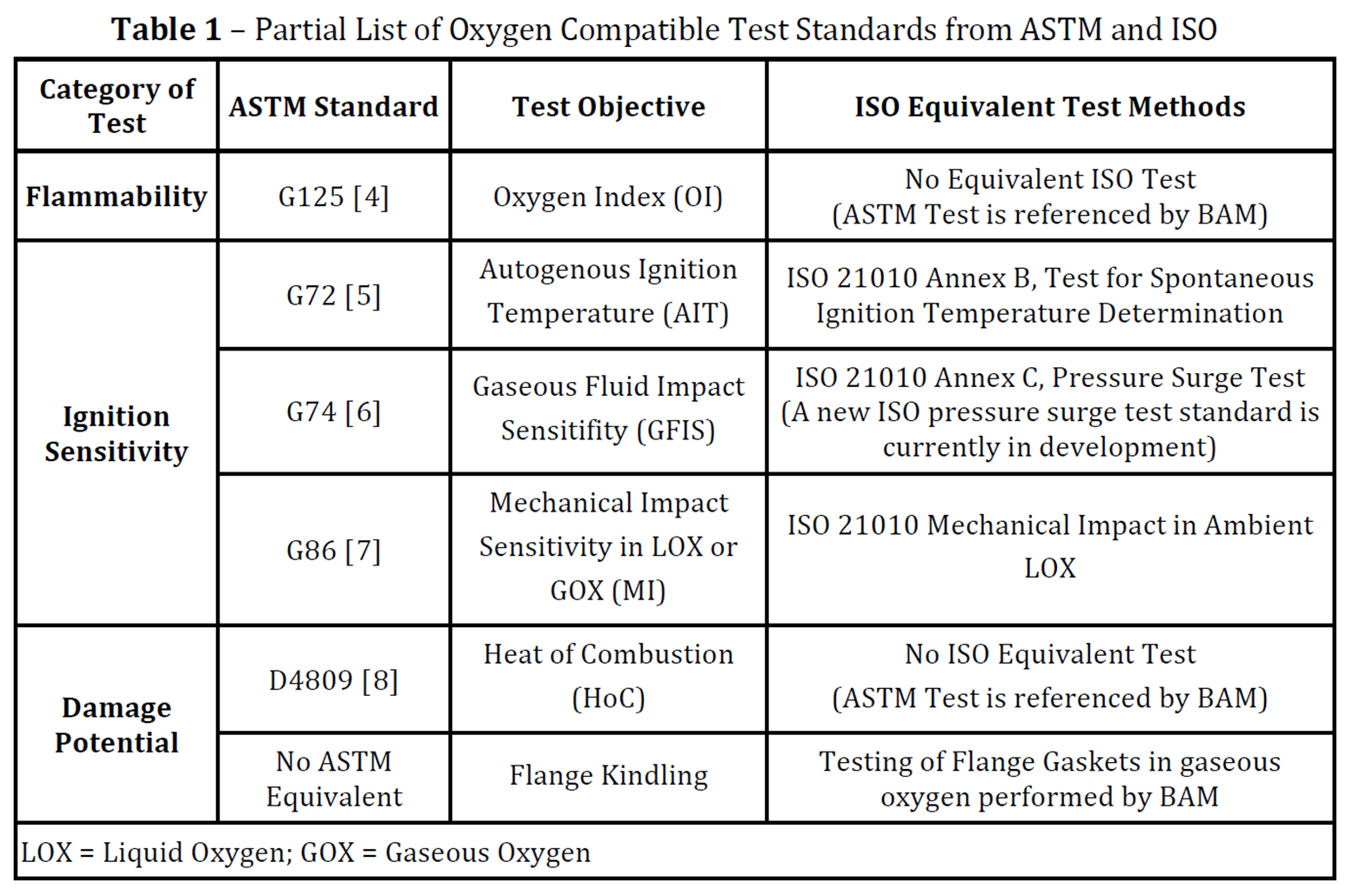
- Flammability tests, which evaluate whether a material will self-sustain burning (i.e. burn on its own once ignited) in a specific configuration under specific conditions (pressure, temperature, concentration). The Oxygen Index test (ASTM G125) evaluates whether a nonmetallic material will self-sustain burning at 1-atm pressure.
- Ignition sensitivity tests, which evaluate ignitability, or the energy required for ignition, of a material in a specific configuration and oxygen test conditions (pressure, temperature, concentration, flow). Multiple tests exist to help us understand the various ways to impart enough energy to ignite a material in oxygen. Such “ignition mechanisms” can include environmental exposure conditions (i.e., autogenous ignition temperature or spontaneous ignition temperature), gaseous fluid impact (i.e., compression heating), and mechanical impact (applied mechanical loading).
- Damage potential tests, which evaluate the energy release by a material that has ignited in oxygen (e.g., heat of combustion). These tests don’t measure the probability of ignition but help evaluate the consequence of a fire once it has started.
Five tests are frequently used by WHA to help assess oxygen compatibility of nonmetals and are listed below in Table 1.
Is there such a thing as oxygen safe-use criteria?
Over many decades of research, testing, and publications, industry can now reference a wealth of information about various materials’ compatibility in oxygen. Although this information provides valuable tools, it has also led to problematic misconceptions.
Such a misconception commonly occurs around a pamphlet published by The Federal Institute for Materials Research and Testing in Germany (BAM). The document, M 034-1 “List of nonmetallic materials compatible with oxygen,” provides a listing of nonmetallic materials based primarily on their behavior in spontaneous ignition testing and pressure surge testing (ISO 21010). The document clearly states that the data found in the document is intended only “to provide preliminary information on oxygen compatibility,” but readers sometimes assume the data to represent universal safe-use criteria for maximum pressure and temperature regardless of application.
Misinterpretation of this data and criteria can lead to dangerous situations. Conditions considered to be “safe” in one setting may pose unacceptable risks in another. We cannot emphasize enough: oxygen compatibility and safe-use criteria depend on application.
What counts as acceptable ignition criteria?
Many people don’t appreciate that all common nonmetals, including plastics, elastomers, and lubricants, are known to be flammable if ignited in pressurized oxygen. In other words, they are all potential fuels if ignited. There are no nonmetals with a zero probability of ignition in pure oxygen. For these reasons, oxygen compatibility and safe-use criteria are based on what is determined to be acceptable risk.
There is a common misperception that a material cannot ignite or burn (i.e. is not flammable) below BAM’s published maximum use pressure and temperature, but this is not true. In general, these conditions are intentionally conservative, but they are not flammability or ignitability limits, and they may have a large uncertainty associated with them. Some of these maximum values are set with as few as two test samples. Such limited data alone cannot provide statistical confidence for unconditional safe-use. In fact, no single material test can “qualify” a nonmetal for unconditional safe-use.
For a given application, the most effective determination of a material’s compatibility is an oxygen fire risk analysis which systematically evaluates a material’s flammability, ignition probability, and ignition consequence. An OFRA uses compatibility test data to defend choices of materials that demonstrate low probability of ignition and fire.
Included in the OFRA are assessments of fire propagation, or kindling chains within a component or system. In other words, once ignited, can one material kindle fire to other materials, and turn a small ignition into a large fire that could cause catastrophic injury or death to personnel?
At WHA, our oxygen safety consultants perform OFRAs and also train others to do so through our Level 3: O2 Design and Level 4: O 2 Analyze oxygen safety training courses.
How can configurational testing support an OFRA?
Although OFRAs provide a very comprehensive analysis of the ignition probability and consequence in a component, they also have their limitations. Sometimes engineers must know more definitively how a material will perform in a specific real-world or in-situ application. Recently, several WHA and Flowserve engineers partnered to do just that by testing a “representative sample” of a larger valve for ignition and fire in oxygen. This work will be published by ASTM in an upcoming peer-reviewed technical paper later this year.
The paper focuses on test standard ISO 21010:2017 and the application of oxygen pressure surge testing on large piping systems. Currently, performing this test on a large industrial valve is not feasible with the test system described in ISO 21010. Component designers therefore have difficulty collecting relevant data based on real world conditions.

The paper’s authors solve this problem with a special fixture designed to emulate use conditions on a smaller scale. Under the new method, thorough oxygen pressure surge testing can be performed on O-rings within a more practical, affordable partial assembly.
You can read more about the background and specifics of this new method in the upcoming ASTM paper “Configurational Oxygen Compatibility Testing of Nonmetals” by Greg Odom, Kevin Smith, Rob Carlson, Gwenael Chiffoleau, and Barry Newton.
We’re here to help simplify oxygen safety
For an in-depth look at these topics, look for “Oxygen Compatibility and Safe-Use Criteria for Non-Metallic Materials” by WHA engineers Barry Newton, Gwenael Chiffoleau, and Elliot Forsyth. This paper will be published soon by ASTM International.
Our experienced oxygen safety consultants are also available to help you understand material compatibility as it applies to your unique oxygen application through standard compatibility testing and Oxygen Fire Risk Analysis (OFRA). You can also learn more about materials compatibility and fire risk analysis in WHA’s technical training courses.
Share this entry
Related Articles
What New CSA Group Research Reveals about Hydrogen Leak Testing Gaps
How do we reliably detect, measure, and manage hydrogen leaks? A new CSA Group research study takes…
Case Study: Steel Mill Industrial Oxygen Fire
In 2013, an industrial oxygen fire tore through a new oxygen supply system feeding an electric arc…
Guide to ASTM G93-2025: A Risk-Based Update to Oxygen Cleanliness
ASTM G93 is a familiar reference for many engineers who work with oxygen systems. It’s often cited…
Request an expert
consultation
Contact us to request a free consultation with an experienced engineer who can help you better understand your needs and our solutions.



